I. BACKGROUND AND HISTORY
Early work in nuclear physics l,2,3,4 indicated that
the nuclear decay parameter, l , was a constant. It was and still
is generally believed that this decay rate is unaffected by changes
in external parameters such as pressure, temperature, electromagnetic
fields, and differing chemical environments. It is believed that
radioactivity is a spontaneous, random event. The fundamental
assumption made is that the decay of an atom is independent of
the age of that atom.5
A brief look at a few nuclear physics texts 6,7,8
shows that at best the precision of nuclear decay parameters is
known only to three significant figures and in some cases to four.
Hence experimental data permits the possibility of a slight variation
in the nuclear decay parameter. This variation could be as much
as one part per thousand per year. Measurement of nuclear disintegration
parameters has been done for about fifty years. To my knowledge
no major research effort has been mounted to determine whether
nuclear decay parameters vary at all with time. Once values of
the decay index for a particular nuclide are obtained and a particular
value is agreed upon, this value is generally accepted.
Usually no further measurements are taken. The terminology is
such that no change with time is expected. Note the quantity,
l , is called the nuclear decay constant. Hence it is easy
to see why a slight change in decay rate, if it exists, could
have been overlooked during the past fifty years.
II. THE CASE FOR CHANGING NUCLEAR DECAY PARAMETER
A. External parameters. External parameters can effect
slight changes in the nuclear decay rate.9 DeYoung
10 has listed variations of up to 5%.
B. Dudley's hypothesis.11 Dudley rejects the
idea that radioactive decay is completely spontaneous and instead
hypothesizes that neutrinos from the neutrino sea, which has been
suggested by others, trigger radioactive decay.
C. Radiohalos. Radiohalos are minute concentric rings
of discoloration in minerals such as mica formed by alpha ray
bombardment from radioactive inclusions. Radiohalos give evidence
that decay rates vary with time.12,13 Some researchers
believe otherwise.
D. The speed of light may have been faster in the past.
Morton has proposed that the permittivity of free space has changed.14
It then follows that the speed of light has changed. Furthermore,
in the case of beta emission, Morton derives a relation between
the nuclear decay parameter and the permittivity. If the permittivity
has changed, it follows that the nuclear decay parameter has changed.
Some investigators claim that there appears to be experimental
evidence that the speed of light decreased in the past although
the speed of light is constant at the present time. If this is
true they argue in the following manner. The energy of emitted
particles from the nucleus is related to the velocity of light
through the relativistic expression for kinetic energy and the
half life of a radioactive atom is related to the energy of the
ejected particle by means of the empirical relation called the
Geiger-Nuttal law. Through these relations we can deduce that
if the speed of light is slowing down, then the radioactive decay
rate is also slowing down.
E. Observed values of half lives. There is a very wide
dynamic range of half lives. The longest are about 109
(one billion) years. Some examples are the half life of U235
which is 0.7 x 109 years, the half life of U238
which is 4.5 x 109 years and the half life of Th232
which is 14.9 x 109 years. At the other extreme examples
of radioisotopes with very short half lives are Po211
which has a half life of 1.64 x 10-4 seconds and At2l6
which has a half life of 3 x 10-4 seconds.
The radioisotope carbon 14 which is used in radiometric dating
has a half life which is not that precisely known. Values ranging
from 5570 to 5730 years have appeared in the literature, with
the larger values being the more recent ones.
Note that the neutron in the environment of the nucleus is a
stable particle, but as a free particle the neutron has a half
life of eight minutes. Thus the environment of the nucleus is
extremely important.
Examination of the published half lives of long-lived radioisotopes
indicates a precision of about one part per thousand. Two cases
where it appears that the half life is increasing with time are
as follows. Glasstone15 (1950) has the half life for
Proactinium 231 as 3.2 x 104 years while Kaplan l6 (1962)
has the half life as 3.43 x 104 years. For the
half life of Radium 223, Glasstone has 11.2 days while Kaplan
has 11.68 days.
III. THEORY
Consider the following three cases: (1) the disintegration index
l , is constant; (2) the disintegration index, l , varies linearly
with time; and (3) the disintegration index, l , varies exponentially
with time.
Each case will result in a corresponding mathematical expression
which can be solved for the number of radioactive atoms present
at a particular time. From these solutions, expressions can be
derived for the half life, t 1 2 . The mathematical
expressions for the previously mentioned quantities are developed
in the Appendix. The constant part of the nuclear decay parameter
is a . Calling this the nuclear "decay constant" the
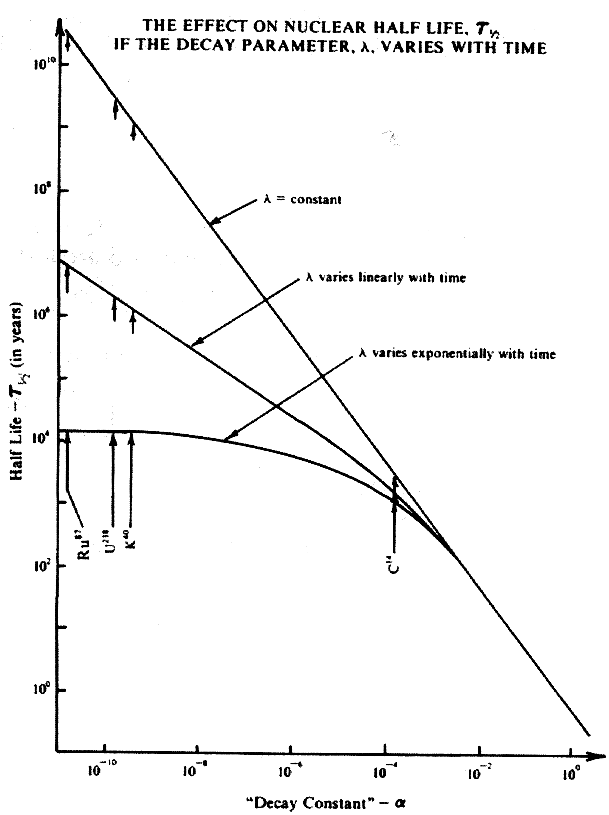
figure is a plot of the half life versus the "decay constant."
The half life of a radionuclide is determined by the time t it
takes for the parent material to be reduced to one half of its
original value. Once the half life is found, users of the radiometric
dating technique, take the expression for half life deduced by
going forward in time and apply it to events which are reached
by going backward in time. One ought to determine the half
life by finding the time required to double the parent material
if one goes backward in time. It is true, of course, that for
a constant disintegration index, the half life computed by both
methods is identical. However, for a variable disintegration index,
the two expressions are significantly different.
IV. ANALYSIS
Plots of equations 3, 8, and 9 of the Appendix are shown of t
1 2 versus a in the figure. The important radionuclides
in radiometric dating are U238, K40, Ru87,
and C14. They are said to have half lives of 4.5 billion
years, 1.2 billion years, 50 billion years, and 5.73 thousand
years respectively. The figure indicates the values for the half
lives for these radionuclides for linear and exponential variation.
The plot shows that for extremely low values of a in the exponential
case, the half lifeis reduced orders of magnitude. For an exponential
variation U238 does not have a half life of 4.5 billion
years but only around 10,000 years. For large values of a , the
half life of the exponential variation is the same as for the
constant case.
Note that b = .001 was used. For smaller values of b , the values
of t 1 2 are the same for even smaller values of a
but when a is small enough, the t 1 2 value for the
exponential variation deviates from the straight line plot and
becomes significantly smaller.
V. CONCLUSION
If a small amount of exponential variation occurs in the nuclear
decay index, then the half lives of the radiometric nuclides are
drastically reduced-orders of magnitude. In the case of U238
the half life is reduced by a factor of 10 5. Hence
the whole area of radiometric dating ought to be rethought.
The reason that there is such a great change in half life due
to a minuscule change in decay index is due to the extrapolation
technique of radiometric dating. When one uses the radiometric
dating technique, one extrapolates from a time base of days at
most to billions of years. The extrapolation factor in time is
approximately 1011. Any competent experimentalist knows
that extrapolation by such a large factor is very poor technique.
Hence one should expect the radiometric dating technique to run
into formidable problems.
There appears to be a difficulty in this approach when one considers
carbon 14. For the linear case the calculated half life is approximately
one half of the currently accepted half life and in the exponential
case it is approximately one third. The carbon 14 method generally
agrees quite well with historical dates. Perhaps the assumed time
dependence should be reduced more�say by a factor of 10. Then
the calculated carbon 14 half life for the exponential case is
only 1.4 times less than the currently accepted value.
The literature should be thoroughly researched for published
values of nuclear decay parameters to determine whether any long-term
variation with time exists. In addition, recent measurements of
half lives should be made and then compared to measurements taken
much earlier in time.
REFERENCES
1. Kaplan, I., Nuclear Physics, Addison-Wesley,
1962, p. 231.
2. Richtmyer, F.K., E.H. Kennard, and J.N. Cooper,
Introduction To Modern Physics, 6th ed., McGraw-Hill,
1969, p. 215.
3. Halliday, D., lntroductory Nuclear Physics,
2nd ed., John Wiley & Sons, 1955, p. 66.
4. Evans, R.D., The Atomic Nucleus, McGraw-Hill,
1955, p. 470.
5. Ibid., p. 471.
6. Kaplan, pp. 248-250.
7. Richtmyer, et al., p. 219.
8. Halliday, p. 64.
9. Anderson, J.L., and G.W. Spangler, "Radiometric
Dating: Is the 'Decay Constant' Constant?", Pensee,
V. 4, No. 4, Fall 1974, p. 31.
10. DeYoung, D.B., "The Precision of Nuclear Decay Rates,"
Creation Research Society Quarterly, V. 13, June 1976,
p. 38.
11. Slusher, Harold S., Critique of Radiometric Dating, ICR
Technical Monograph No. 2, 2nd ed., 1981, p. 20.
12. Ibid., pp. 25-26.
13. Gentry, R.V., "On the lnvariance of the Decay Constant
Over Geologic Time," Creation Research Society
Quarterly, V. 5, No. 2, 1968, p. 83.
14. Morton, G.R., "Electromagnetics and the Appearance
of Age," Creation Research Society Quarterly, March
1982.
15. Glasstone, S., "The Sourcebook on Atomic
Energy," Van Nostrand, 1950, pp. 125-128.
16. Kaplan, p. 249.












