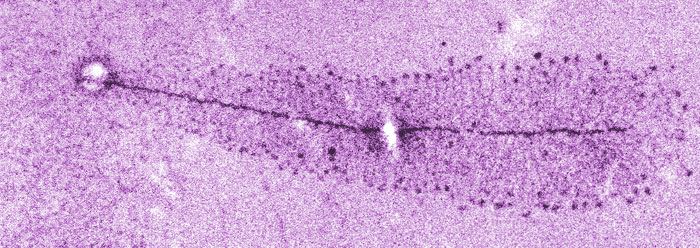
Researchers at Wayne State University in Michigan have uncovered a key step in the formation of antibodies. It was already known that the immune system generates a variety of antibodies in response to an invading pathogen. The recent study discovered that many of the necessary antibody variations are produced when a cellular copying procedure is slowed down.1
Antibodies are manufactured with variations on one end, the “light chain” end. When a specific light chain variation is found that locks onto the outermost molecules of the invading bacterium or virus, the antibody marks the microbe for destruction. The correctly-fitting antibody is then identified and mass-produced by the immune system.
Antibody light chain variation occurs through the acceleration of mutations. The antibody genes are copied to RNA and a special enzyme called “activation-induced deaminase” encourages the shuffling of just the base sequences that will become the light chain. The new research demonstrated that when transcription is slowed down—a process called RNA transcription stalling—this base-exchanging process moves from just one base at a time to multiple bases. This “hypermutation” causes faster production of antibody variations, thus enhancing their disease-combating potential.
Though this discovery was hailed as “evolution in action,” the antibody formation procedures show distinct marks of intentional design.2 Creation scientists have described the capacity of antibodies to hypermutate as a well-designed and purposeful biological process.3 This interpretation is borne out by the specifications of the process, each of which represents information that nature alone has no mechanism to generate. The three-dimensional structure of activation-induced deaminase, the placement of mutations (RNA base changes) specifically on the tips of the antibodies, and the placement of immunoglobulin genes into appropriate segments of the genome so they can be transcribed together are some of the specifications required for antibody variations to effectively achieve their purposes.
The term “evolution” can be used to describe any change in biological systems, or it can refer specifically to Darwinian evolution. However, a step-by-step progression from simple to complex living organisms, as Darwinian evolution predicts, is not observed in either living or fossil forms. Nor does antibody variation have anything whatever to do with Darwinian evolution. In actuality, biological adaptations such as antibody diversity are created mechanisms that exist for specific purposes and only work because all of their parts are precisely ordered.
The variety of antibodies that can be formed is an important part of the immune system’s ability to detect and fight disease. The variety of definitions under the banner “evolution,” however, is often only effective in providing false confidence that “Darwinian evolution” is true.
References
- Canugovi, C., M. Samaranayake, and A. S. Bhagwat. 2009. Transcriptional pausing and stalling causes multiple clustered mutations by human activation-induced deaminase. The Journal of the Federation of American Societies for Experimental Biology. 23 (1): 33-44.
- Evolution in action: Our antibodies take “evolutionary leaps” to fight microbes. The FASEB Journal press release, accessed January 12, 2009.
- Bergman, J, and N. O’Sullivan. 2008. Did immune system antibody diversity evolve? Journal of Creation. 22 (2): 92-96.
* Mr. Thomas is Science Writer.
Article posted on January 21, 2009.