According to the neo-Darwinian interpretation of evolution, all living forms have arisen from a single form of life by slow gradual changes. Thus, the time between the origin of life and the abrupt appearance in the fossil record of the many complex invertebrate forms of life is now estimated to have been nearly three billion years. The time required for one of these invertebrates to evolve into the vertebrates, or fishes, has been estimated at about 100 million years, and it is believed that the evolution of the fish into an amphibian required about 30 million years. The essence of the neo-Darwinian view is the slow gradual evolution of one plant or animal into another by the gradual accumulation of micromutations through natural selection of favored variants.
If this view of evolution is true, the fossil record should produce an enormous number of transitional forms. Natural history museums should be overflowing with undoubted intermediate forms. About 250,000 fossil species have been collected and classified. These fossils have been collected at random from rocks that are supposed to represent all of the geological periods of earth history. Applying evolution theory and the laws of probability, most of these 250,000 species should represent transitional forms. Thus, if evolution theory is true, there should be no doubt, question, or debate as to the fact of evolution.
Such is not the case at all, however. The fossil record was actually an embarrassment to Darwin, and some paleontologists are willing to admit that it looks even worse from an evolutionary point of view today than it did in Darwin's time.1 Some even appear to admit that there is, in fact, little, if any, evidence for transitional forms in the fossil record. Kitts, for example, states, "Despite the bright promise that paleontology provides a means of 'seeing' evolution, it has presented some nasty difficulties for evolutionists, the most notorious of which is the presence of 'gaps' in the fossil record. Evolution requires intermediate forms between species and paleontology does not provide them." More and more paleontologists seem to be coming to the point where they are now willing to admit that this is indeed the case, and are seeking to devise a mechanism for evolution that will tolerate, even predict, systematic gaps in the fossil record.
Other evolutionists remain steadfastly wedded to neo-Darwinism. They argue that there are examples of transitional forms in the fossil record, and that even if examples of gradual change are few, these few examples eliminate the necessity of seeking mechanisms for evolution other than neo-Darwinism. The examples most often cited are the reptile-to-bird transition (Archaeopteryx is the sole suggested intermediate), the so-called horse series, and the reptile-mammal transition.
Of the latter, Olson has said "The reptilian-mammalian transition has by far the finest record of showing the origin of a new class."2 Others claim that there are forms that stand perfectly on the reptilian-mammalian boundary. In an "Impact" article to be published soon, we will examine in some detail the "mammal-like" reptiles that some paleontologists believe represent transitional forms between reptiles and mammals. In the present article we wish to review the general nature of the evidence related to the origin of mammals.
The creatures included within the class Mammalia are a diverse group. All are warm-blooded and the females possess mammary glands for suckling the young. The mammals comprise 32 orders, most of which are placental mammals but which also include the Monotremata, which embrace the egg-laying spiny ant-eater and the egg-laying duckbilled platypus, and the Marsupialia, which include the opossums and the pouched marsupials, such as the kangaroo and wallabies.
It is interesting to note that while claiming that intermediate forms for the reptile-to-mammal transition have been found, some evolutionists admit that no immediate ancestors for any of the 32 mammalian orders have been discovered. Thus, George Gaylord Simpson, after stating that nowhere in the world is there any trace of a fossil that would close the considerable gap between Hyracotherium ("Eohippus"), which evolutionists assume was the first horse, and its supposed ancestral order Condylarthra, goes on to say "This is true of all the thirty-two orders of mammals…The earliest and most primitive known members of every order already have the basic ordinal characters, and in no case is an approximately continuous sequence from one order to another known. In most cases the break is so sharp and the gap so large that the origin of the order is speculative and much disputed."3
The marine mammals thus abruptly appear in the fossil record as whales, dolphins, sea-cows, etc. For example, in one of Romer's concluding statements in his discussion of the subungulates (conies, elephants, sea-cows), he says "… conies, proboscideans, and sirenians were already distinct groups at the time when they first appear in the fossil record."4 Olson states that if we seek the ancestries of the marine mammals we run into a blank wall as far as intermediate stages between land and sea are concerned.5 His remark included the seals, dolphins and whales. There simply are no transitional forms in the fossil record between the marine mammals and their supposed land mammal ancestors.
Romer suggests that whales may have descended from a primitive carnivore,6 although concerning the Sirenia (sea cows) and Cetacea (whales, dolphins) he admits that "We are ignorant of their terrestrial forebears and cannot be sure of their place of origin."7 It is interesting to note that many of the so-called "primitive" carnivorous mammals had about 40 teeth differentiated into incisors, canines, premolars and molars. The porpoises, dolphins, and whales, however, may possess teeth far in excess of that number (one porpoise has 300), and the teeth of these marine mammals are usually simple pegs or wedges and are not differentiated into incisors, canines, premolars and molars.
Würsig has suggested, on the other hand, that dolphins may have evolved from land mammals resembling the even-toed ungulates of today such as cattle, pigs and buffaloes.8 It is quite entertaining, starting with cows, pigs, or buffaloes, to attempt to visualize what the intermediates may have looked like.
Bats (of the order Chiroptera), the only flying mammal, are especially interesting. Evolutionists assume, of course, that bats must have evolved from a non-flying mammal. There is not one shred of evidence in the fossil record, however, to support such speculations, for, as Romer says, "Bats appear full fledged in both hemispheres in the Middle Eocene …"9
On the cover page of Science of December 9, 1966 (Vol. 154) appears a picture of what the author (Glenn L. Jepsen) of the accompanying article (pp. 1333-1339) describes as the oldest known bat. He reports that it was found in Early Eocene deposits, which are dated by evolutionists at about 50 million years. While stating that this bat possessed a few "primitive" characteristics, Jepsen states that it was fully developed, an "anatomically precocious" contemporary of Eohippus. Thus, bats appear fully-formed, with no trace of ancestors or intermediate forms, as a contemporary of Eohippus, supposedly the ancestor of horses. According to Jepsen this leaves many questions unanswered, including when, from what, where, and how did bats originate?
Horses comprise one of the most interesting mammalian groups as far as the question of origins is concerned. Almost all students are familiar with the story of horse "evolution," beginning with Hyracotherium (Eohippus), a dog-sized "horse" with four toes on the front feet, passing via straight-line evolution through three-toed varieties, and ending with the modern one-toed Equus. But while subscribing to the evolution of the horse in general, Birdsell proclaims that "Much of this story is incorrect …"10 Others hold the same view. George Gaylord Simpson, for example, has declared that several generations of students have been misinformed about the real meaning of the evolution of the horse.11 These authors believe that the evolution of the horse is much more complicated than usually portrayed, and is more like a series of bushes, perhaps, than like a tree.
To us the family tree of the horse appears to be merely a scenario put together from non-equivalent parts. Nowhere, for example, are there intermediate forms documenting transition from a non-horse ancestor (supposedly a condylarth) with five toes on each foot, to Hyracotherium with four toes on the front foot and three on the rear. Neither are there transitional forms between the four-toed Hyracotherium and the three-toed Miohippus, or between the latter, equipped with browsing teeth, and the three-toed Merychippus, equipped with high-crowned grazing teeth. Finally, the one-toed grazers, such as Equus, appear abruptly with no intermediates showing gradual evolution from the three-toed grazers.
 |
Thus, Birdsell tells this story in the following way (note that when an evolutionist uses such terms as "sudden," "abrupt," or "rapid" with reference to transitions he is usually inferring that no transitional forms have been found): "The evolution of the foot mechanisms proceeded by rapid and abrupt changes rather than gradual ones. The transition from the form of foot shown by miniature Eohippus to larger consistently three-toed Miohippus was so abrupt that it even left no record in the fossil deposits… their foot structure [those of Miohippus] changed very rapidly to a three-toed sprung foot in which the pad disappeared and the two side toes became essentially functionless. Finally, in the Pliocene the line leading to the modern one-toed grazer went through a rapid loss of the two side toes on each foot."12 He then goes on to say that this evolution was not gradual but that it had proceeded by rapid jumps. Thus, the continuity required by theory cannot be documented from the fossil record.
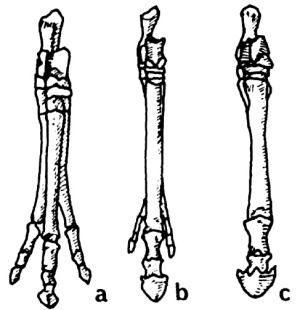
A rather astounding and revealing fact is discovered when we compare North American ungulates to South American ungulates. All of us are familiar with the series shown in Fig. 1. These are the front feet (pes) of, a, "Eohippus"; b, Merychippus, with reduced lateral toes; and c, modern Equus. Now look at Fig. 2. Illustrated are the pes of the South American ungulates (order Litopterna), a, Macrauchenia; b, Diadiaphorus; and c, Thoatherium. Again we see a three-toed hoofed ungulate (Macrauchenia); a three-toed hoofed ungulate with reduced laterals (Diadiaphorus); and, in this case, a one-toed hoofed ungulate (Thoatherium) which, Romer says, seems even more horselike than any true horse, for it was single-toed with splints more reduced than those of modem equids.13
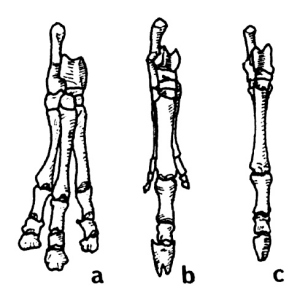 |
Do they not thus provide another nice, logical evolutionary series? No, not at all, for they do not occur in this sequence at all! Diadiaphorus, the three-toed ungulate with reduced lateral toes, and Thoatherium, the one-toed ungulate, were contemporaries in the Miocene epoch. Macrauchenia, with pes containing three full-sized toes, is not found until the Pliocene epoch, which followed the Miocene according to the geological column. In fact, it is said that the one-toed Thoatherium became extinct in the Miocene before the three-toed Macrauchenia made his appearance in the Pliocene.
Thus, if evolutionists would permit the fossil evidence and their usual assumptions concerning geological time to be their guide, they should suppose that in South America a one-toed ungulate gave rise to a three-toed ungulate with reduced lateral toes, which then gave rise to an ungulate with three full-sized toes. This is precisely the opposite of the supposed sequence of events that occurred with North American horses. I don't know any evolutionist who suggests such an evolutionary sequence of events, but why not? Perhaps it is because the three-toed to one-toed sequence for North American horses became so popularized in evolutionary circles that no one dare suggest the reverse transition. Of course there is no more real evidence for transitional forms in South America than there is in North America.
It should also be noted that in the Rattlesnake Formation of the John Day Country of northeastern Oregon, the three-toed horse Neohipparion is found with the one-toed horse, Pliohippus.14 No transitional forms between the two are found. In other cases "primitive" species of a genus, such as those of Merychippus, are found in geological formations supposedly younger than those containing "advanced" species.15
Was Hyracotherium (Eohippus) really a horse? Hyracotherium was discovered in Europe before "Eohippus" was uncovered in North America, and was given the genus designation of Hyracotherium by the famous British anatomist and paleontologist, Richard Owen, who was also its discoverer. Later, other specimens were discovered in North America and given the genus name Eohippus. It was subsequently concluded that the North American specimens were actually of the same genus as Hyracotherium. The latter thus has priority, so Eohippus is not a valid name for these creatures. It is most commonly used, however, undoubtedly because the name Eohippus means "dawn horse" while Hyracotherium was chosen by Owen because of the resemblance of this creature to creatures of the genus Hyrax (cony, daman).
Although Hyracotherium, or Eohippus, was unlike modern horses, both morphologically and in habitat, this creature was chosen to stand at the base of the horses by the American paleontologist Marsh, and others, and this scheme became solidly entrenched both in popular circles and in scientific status after a lecture in New York City by Thomas H. Huxley and the publication of Marsh's studies.16
Nilsson has pointed out that while Hyracotherium has little or no resemblances to horses, it apparently was morphologically and in habitat similar to living creatures of the genus Hyrax.17 Hyrax, like Hyracotherium, is about the size of a rabbit or fox. Hyrax, also like Hyracotherium, has four toes on the front feet and three on the rear. The cheek teeth of these two creatures share many similarities and are more like those of rhinoceri than those of horses. The habitat and way of life of Hyrax are also similar to those postulated for Hyracotherium. Thus, Nilsson maintains, although Hyracotherium does not resemble present-day horses in any way, they were, apparently, remarkably similar to the present-day Hyrax.
Others also doubt whether Hyracotherium was related to the horse. For example, Kerkut states, "In the first place it is not clear that Hyracotherium was the ancestral horse. Thus Simpson (1945) states, ‘Matthew has shown and insisted that Hyracotherium (including Eohippus) is so primitive that it is not much more definitely equid than tapirid, rhinocerotid, etc., but it is customary to place it at the root of the equid group.’"18 In other words, Hyracotherium is not any more like a horse than it is similar to a tapir or a rhinoceros, and thus just as justifiably it could have been chosen as the ancestral rhinoceros or tapir. It seems, then, that the objectivity of those involved in the construction of the phylogenetic tree of the horse was questionable from the very start, and that the "horse" on which the entire family tree of the horse rests was not a horse at all.
No definitive work on horses has been published since the publication of Kerkut's book that would materially affect his conclusion that "In some ways it looks as if the pattern of horse evolution might be even as chaotic as that proposed by Osborn (1937, 1943) for the evolution of the Proboscidea, where "in almost no instance is any known form considered to be a descendant from any other known form; every subordinate grouping is assumed to have sprung, quite separately and usually without any known intermediate stage, from hypothetical common ancestors in the Early Eocene or Late Cretaceous' (Romer 1949)."18 If indeed "horse evolution" is that chaotic and patchy, this classic case for evolution is without real merit. The actual evidence, on the other hand, neatly fits the Creation Model.
The order Rodentia should provide evolutionists with a group of mammals ideal for documenting evolutionary transitions. In number of species and genera, the rodents exceed all other mammalian orders combined. They flourish under almost all conditions. Surely, if any group of animals could supply transitional forms, this group could.
As to their origin, Romer has said "The origin of the rodents is obscure. When they first appear, in the late Paleocene, in the genus Paramys, we are already dealing with a typical, if rather primitive, true rodent, with the definitive ordinal characters well developed. Presumably, of course, they had arisen from some basal, insectivorous, placental stock; but no transitional forms are known."19
Furthermore, transitional forms between the basic rodent types are not found in the fossil record. For example, Romer says "… the beavers are presumably derived from some primitive sciuromorph stock, but there are no annectant types between such forms and the oldest Oligocene castoroids to prove direct relationship."19
Speaking of the Hystricidae, the Old World porcupines, Romer says "There are a few fossil forms, back to the Miocene and possibly late Oligocene, but these give no indication of relationship of hystricids to other rodent types."
Commenting on the "rock rat," Petromus, Romer says "Almost nothing is known of the ancestry or possible descendants." Of the lagomorphs (hares and rabbits), once placed in a suborder of the rodents, but now placed in a separate order, Lagomorpha, Romer must admit that, "The lagomorphs show no close approach to other placental groups, and the ordinal characters are well developed in even the oldest known forms."
Thus we see that the order Rodentia, which should supply an excellent case for evolution, if evolution really did occur, offers powerful evidence against the evolutionary hypothesis.
The order Primates is of special interest since that is the order within which our own species, Homo sapiens, is placed. We do not consider the order Primates to be a natural group at all, since prosimians, monkeys, apes, and men each were separately created and did not share a common ancestor. Evolutionists, on the other hand, believe that all of these creatures have shared a common ancestor, and that they shared this common ancestry more recently than they shared their common ancestry with any other animal.
The primates are supposed to have evolved from an insectivorous ancestor, more particularly a creature resembling the tree shrew. There is no evidence whatever in the fossil record, however, to support such an idea, for no transitional forms can be found. Elwyn Simons, a leading evolutionary paleoanthropologist, admits that "In spite of recent finds, the time and place of origin of order Primates remains shrouded in mystery."20 Romer states that the early lemurs (lemur-like creatures were supposed to be among the first primates) appear "apparently as immigrants from some unknown area."21 He suggests this since paleontologists have no indication in the fossil record how lemurs (and thus the primates) arose. Kelso states "… the transition from insectivore to primate is not documented by fossils. The basis of knowledge about the transition is by inference from living forms."22
It is thus evident that when evolutionists seek for the origin of the primate order in the fossil record they encounter a blank wall. Furthermore, recent studies appear to invalidate earlier conclusions that tree shrews (tupaiids) are closely related to living primates. Campbell, who has reviewed this recent work, states "I have attempted to indicate the large number of recent studies where results indicate that a close relationship between tupaiids and primates is unlikely."23 He then goes on to say that the innate attractiveness of including the tree shrew in the sequence tree shrew-lemur-tarsier-ape-man may have been in large measure responsible for its acceptance.
We see, then, that there is no evidence to link the primates to the tree shrews (or to any other supposed ancestor), either in the fossil record or among living creatures. The primates stand distinctly apart from all other groups.
This review, certainly brief and incomplete, should nevertheless be sufficient to document the fact that mammals cannot be linked to reptiles or to any other group. Since it can be shown that each of the 32 orders of mammals are separate and distinct groups set apart from one another and from all other creatures by unbridged gaps, it seems evident that collectively as mammals they are set apart as well.
REFERENCES
1 Raup, David M., Field Museum of Natural History Bulletin, Vol. 50, 1979, p. 25.
2 Olson, E. C., The Evolution of Life, New York: The New American Library, 1965, p. 207.
3 Simpson, G. G., Tempo and Mode in Evolution, New York: Columbia University Press, 1944, p. 105.
4 Romer, A. S., Vertebrate Paleontology, 3rd Ed., Chicago: Univ. of Chicago Press, 1966, p. 254.
5 Olson, E. C., p. 178.
6 Romer, A. S., p. 297.
7 Romer, A. S., p. 339.
8 Würsig, B., Scientific American, Vol. 240, 1979, p. 136.
9 Romer, A. S., p. 338.
10 Birdsell, J. B., Human Evolution, Chicago: Rand McNally College Pub. Co., 1975, p. 169.
11 Simpson, G. G., The Major Features of Evolution, New York: Columbia Univ. Press, 1953, p. 259.
12 Birdsell, J. B., p. 170.
13 Romer, A. S., pp. 260-261.
14 Nevins, S., Creation Research Society Quarterly, Vol. 10, 1974, p. 196.
15 Gregory, J. T., University of California Publications in Geological Sciences, Vol. 26, 1942, p. 428.
16 Cousins, F. W., Creation Research Society Quarterly, Vol. 7, 1971, p. 102.
17 Nilsson, H., Synthetische Artbildung, Verlag CWE Gleerup, Lund, Sweden, 1954. (See F.W. Cousins, Ref. 16, for a summary on the horse.)
18 Kerkut, G. A., Implications of Evolution, New York: Pergamon Press, 1960, p. 149.
19 Romer, A. S., p. 303. The following quotes from Romer are in the section pp. 304-31 0.
20 Simons, E. L., Annals New York Academy of Sciences, Vol. 167, 1969, P. 319.
21 Romer, A. S., p. 218.
22 Kelso, A. J., Physical Anthropology, 2nd Ed., New York: J. B. Lippincott, 1974, p. 142.
23 Campbell, C.B.G., Science, Vol. 153, 1966, P. 436.
* Dr. Duane T. Gish was the Vice President of the Institute for Creation Research. He is the author of several other related articles and books and was in demand as a speaker on scientific creationism throughout the world.



