Within the ever-expanding theory of evolution, there is a system of specialized language designed to identify each major interpretative concept. Some terminology is so versatile that it could be applied to a unique observation or a broad set of patterns across extensive scales of time and space. Such terms appear to be intentionally flexible, or even modifiable. From the introduction of a prestigious book of essays, Keywords in Evolutionary Biology, the authors state,
Within the ever-expanding theory of evolution, there is a system of specialized language designed to identify each major interpretative concept. Some terminology is so versatile that it could be applied to a unique observation or a broad set of patterns across extensive scales of time and space. Such terms appear to be intentionally flexible, or even modifiable. From the introduction of a prestigious book of essays, Keywords in Evolutionary Biology, the authors state,
Our goal in this book has been to identify and explicate those terms in evolutionary biology that, though commonly used, are plagued in their usage by multiple concurrent and historically varying meanings.1
The term “convergent” has varying meanings within evolutionary biology. Definitions include the evolution of similar features in independent lineages;2 similarity between two organisms, structures, or molecules by independent evolution along similar lines;3 and similarity between species caused by similar but independent evolutionary responses to a common environmental problem.4 When considered as a group, these definitions broadly define convergent evolution as showing similar forms and functions in different species that do not descend from a common ancestor. But is convergent evolution actually an observable process?
Compensatory Adaptations in A. mexicanus

At the Institute for Creation Research, we are investigating the development and diversity of adaptations in Astyanax mexicanus (Mexican tetra), a freshwater fish that exhibits two contrasting morphotypes: eyed surface-dwelling fish (surface fish) with distinct pigmentation patterns and eyeless cave-dwelling fish (cavefish) with minimal pigmentation (Figure 1). At least 30 genetically identified populations of A. mexicanus have been recorded from northeastern Mexico.5 All cavefish strains share a similar set of highly optimized adaptations to subterranean environments (caves), which are interpreted by most conventional scientists as having evolved by convergent evolution.6–8 However, all A. mexicanus cavefish and surface fish are members of a single species, and thus an interpretation of convergence is problematic.
By direct comparison with the eyed surface-dwelling form, eyeless cavefish exhibit specific adaptations that compensate for an absence of vision. There are enhancements in feeding, smell, respiration, energy storage and metabolism, prey capture, and navigation.9,10 For example, cavefish develop larger jaws with more teeth, increased numbers of oral taste buds, and a wider distribution of extraoral taste buds with age.11 These feeding-related (gustatory) adaptations are likely correlated with enlargement of the hypothalamus, its associated neuroanatomy,12 and the “greatly enhanced” circuitry of larger olfactory pits, bulbs, and epithelia with higher chemosensory capabilities in cavefish.12–14 During development, embryonic olfactory tissues (nasal placodes) are comparatively larger in the cavefish, resulting in larger olfactory epithelia and nerves with an ability to detect 105-fold lower concentrations of amino acids than surface fish of similar age.14
Respiratory compensations include smaller hearts with rounded chambers (ventricles) and more muscular ridges (trabeculae), providing a “larger surface area” for higher oxygen uptake.15 And although cavefish possess fewer red blood cells (erythrocytes) than surface fish, the two-dimensional surface area of those cells is “significantly larger.”16 This indicates a higher concentration of hemoglobin per erythrocyte and measurably higher concentrations of blood hemoglobin when compared with conspecific surface fish. This is a critical compensation for life under low-oxygen conditions commonly found within karst cave environments.
As for energy storage and utilization within nutrient-poor habitats, cavefish increase their foraging behavior and offset starvation periods from larval through adult stages by continuous formation of fat cells (adipocytes) to compensate for food scarcity.17 They have comparatively more and larger adipocytes than surface fish and accumulate triglycerides that enable them to survive on fatty acids.18,19 In fact, cavefish have hyperglycemia, insulin resistance, elevated triglyceride levels, and fatty livers without harmful pathologies.20,21 Metabolic conditions that would otherwise be detrimental are instead integrated for energy efficiency in caves.
Sensory Compensation for Vision Loss
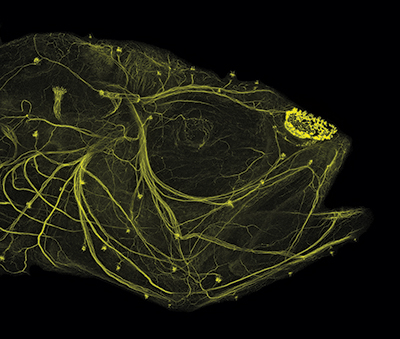
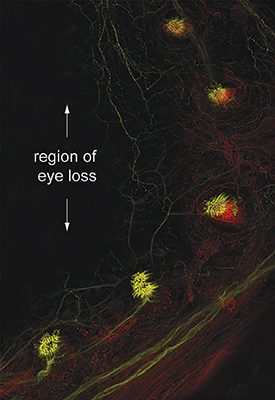
One of the most critical adaptations for cavefish survival involves expansion of the lateral line.23 Almost all fish have a lateral line system of neuromasts (sensors) that detect water flow and vibration. Each neuromast contains an array of mechanosensory hair cells that transform mechanical signals into chemical signals along nerves (Figure 2). These signals are converted into electrical impulses within the central nervous system.24 Thus, hair cells on the body and head (Figures 3 and 4) transfer sensory input to the brain, which relays that information back to tissues and muscles along the body (e.g., jaws, fins, tail).
Compared with surface fish, both juvenile and adult cavefish possess several-fold more superficial neuromasts across their head region (anterior lateral line),23,25,26 and both canal and superficial neuromasts are larger and contain more sensory hair cells.25 These enhancements are directly linked to vibrational attraction behavior observed in cavefish that swim “toward the source of a water disturbance” in dark cave habitats.26
Cavefish rely upon their lateral line for locating mates during reproduction, avoiding obstacles, and capturing prey.26,27 Even more fascinating, neuromasts develop on the larval head and trunk prior to degeneration of the retina.28,29 This implies that the timing of early neuromast development “anticipates” the provision of adaptive sensory compensation for the loss of vision in A. mexicanus larvae.
Cavefish are certainly not the only cave-dwelling animals. Other troglomorphs with similar trait adaptations include crickets, spiders, centipedes, flatworms, snails, salamanders, and more. From a conventional worldview, similar traits across a diversity of body plans would actually support the interpretation of evolutionary convergence—similar responses to a common environment among taxa with uncommon ancestry.
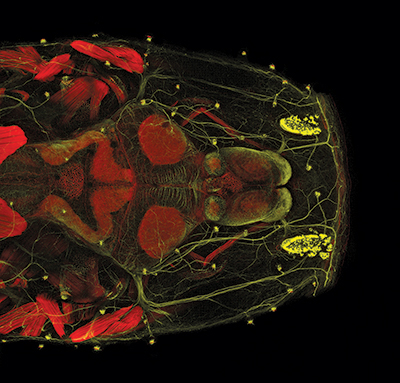
But remember, A. mexicanus cavefish and surface fish are members of an interbreeding group of fish. They are the same species! This is stunning since the conventional cavefish community suggests that regressive changes (loss of eyes, pigmentation, schooling, aggression, circadian rhythm) and constructive changes (jaws, teeth, tastebuds, olfactory bulbs, fat storage, insulin resistance, lateral line expansion) within A. mexicanus also support convergent evolution.9,10 Whether cavefish diverged from surface fish millions of years ago8 or only several thousand years ago,30 all varieties of A. mexicanus comprise one species, which by definition could never arise by convergent evolution. They can’t have it both ways!
There is another explanation. At ICR, we hold a unique view on the origin and deployment of adaptations.6 Within Astyanax cavefish, there is an integrated system of preprogrammed adjustments that actively deploy in response to stimuli in subterranean environments. As with all other organisms, these fish continuously track a range of environmental parameters, assess those parameters on multiple levels (e.g., biochemical, genetic, cellular, physiological), and self-adjust. We model this process as continuous environmental tracking (CET).31
Divine Engineering
We predict that essential adaptations in cavefish and a broad diversity of unrelated cave-adapted (troglomorphic) body plans will become functional within one or two generations after their initial entrance into limestone cave environments. Accordingly, their anatomical, physiological, and behavioral adjustments will be rapid, repeatable, and reversable. In every case, the organism is in control of every adaptive change; the environment (nature) has no inventive or creative power.
Thus, there is one Creator, the Lord Jesus, who has endowed all organisms with such potential, providing the most authoritative explanation for how and why cavefish exhibit nearly identical functionality worldwide.32 The myth of convergent evolution is therefore unmasked by the divine engineering of developmentally integrated solutions to similar environmental challenges. There is only one Engineer who has the power to create such adaptive potential. “For since the creation of the world His invisible attributes are clearly seen, being understood by the things that are made.” (Romans 1:20).
References
- Keller, E. F. and E. A. Lloyd. 1992. Keywords in Evolutionary Biology. Cambridge, MA: Harvard University Press, 3–4.
- Urry, L. A. et al. 2017. Campbell Biology. London, UK: Pearson Education, Inc.
- Lawrence, E. 2016. Henderson’s Dictionary of Biology, 16th ed. London, UK: Pearson Education, Inc.
- Herron, J. C. and S. Freeman. 2014. Evolutionary Analysis, 5th ed. London, UK: Pearson Education, Inc.
- Warren, W. C. et al. 2021. A Chromosome-Level Genome of Astyanax mexicanus Surface Fish for Comparing Population- Specific Genetic Differences Contributing to Trait Evolution. Nature Communications. 12, article 1447.
- Boyle, M. J. et al. 2023. Testing the Cavefish Model: An Organism-Focused Theory of Biological Design Biological Design. Proceedings of the International Conference on Creationism 9, article 78: 654.
- Wilkens, H. and U. Strecker. 2003. Convergent Evolution of the Cavefish Astyanax (Characidae, Teleostei): Genetic Evidence from Reduced Eye-Size and Pigmentation. Biological Journal of the Linnean Society. 80 (4): 545–554.
- Gross, J. B. 2012. The Complex Origin of Astyanax Cavefish. BMC Evolutionary Biology. 12, article 105: 1–12.
- Jeffery, W. R. 2020. Astyanax Surface and Cave Fish Morphs. EvoDevo. 11, article 14.
- Jeffery, W. R. 2001. Cavefish as a Model System in Evolutionary Developmental Biology. Developmental Biology. 231 (1): 1–12.
- Berning, D., H. Heerema, and J. B. Gross. 2024. The Spatiotemporal and Genetic Architecture of Extraoral Taste Buds in Astyanax Cavefish. Communications Biology. 7, article 951.
- Rétaux, S., K. Pottin, and A. Alunni. 2008. Shh and Forebrain Evolution in the Blind Cavefish Astyanax mexicanus. Biology of the Cell. 100 (3): 139–147.
- Rétaux, S. and D. Casane. 2013. Evolution of Eye Development in the Darkness of Caves: Adaptation, Drift, or Both? EvoDevo. 4 (26): 1–12.
- Hinaux, H. et al. 2016. Sensory Evolution in Blind Cavefish Is Driven by Early Embryonic Events during Gastrulation and Neurulation. Development. 143 (23): 4521–4532.
- Tang, J. L. et al. 2018. The Developmental Origin of Heart Size and Shape Differences in Astyanax mexicanus Populations. Developmental Biology. 441 (2): 272–284.
- Boggs, T. E., J. S. Friedman, and J. B. Gross. 2022. Alterations to Cavefish Red Blood Cells Provide Evidence of Adaptation to Reduced Subterranean Oxygen. Scientific Reports. 12, article 3735.
- Xiong, S. et al. 2018. Early Adipogenesis Contributes to Excess Fat Accumulation in Cave Populations of Astyanax mexicanus. Developmental Biology. 441 (2): 297–304.
- Medley, J. K. et al. 2022. The Metabolome of Mexican Cavefish Shows a Convergent Signature Highlighting Sugar, Antioxidant, and Ageing-Related Metabolites. Elife. 11: e74539.
- Olsen, L., E. Thum, and N. Rohner. 2021. Lipid Metabolism in Adaptation to Extreme Nutritional Challenges. Developmental Cell. 56 (10): 1417–1429.
- Riddle, M. R. et al. 2018. Insulin Resistance in Cavefish as an Adaptation to a Nutrient-Limited Environment. Nature. 555 (7698): 647– 651.
- Krishnan, J. et al. 2022. Liver-Derived Cell Lines from Cavefish Astyanax mexicanus as an In Vitro Model for Studying Metabolic Adaptation. Scientific Reports. 12, article 10115.
- The raw images for Figures 2–4 were produced in The William B. Dean, MD Imaging Center of the Institute for Creation Research.
- Rodriguez-Morales, R. 2024. Sensing in the Dark: Constructive Evolution of the Lateral Line System in Blind Populations of Astyanax mexicanus. Ecology and Evolution. 14 (4): e11286.
- D’Angelo, L. et al. 2016. Anatomical Features for the Adequate Choice of Experimental Animal Models in Biomedicine: I. Fishes. Annals of Anatomy-Anatomischer Anzeiger. 205: 75–84.
- Lunsford, E. T. et al. 2022. Evolutionary Convergence of a Neural Mechanism in the Cavefish Lateral Line System. Elife. 11: e77387.
- Yoshizawa, M. et al. 2010. Evolution of a Behavioral Shift Mediated by Superficial Neuromasts Helps Cavefish Find Food in Darkness. Current Biology. 20 (18): 1631–1636.
- Lloyd, E. et al. 2018. Evolutionary Shift Towards Lateral Line Dependent Prey Capture Behavior in the Blind Mexican Cavefish. Developmental Biology. 441 (2): 328–337.
- Alunni, A. et al. 2007. Developmental Mechanisms for Retinal Degeneration in the Blind Cavefish Astyanax mexicanus. Journal of Comparative Neurology. 505 (2): 221–233.
- Sarrazin, A. F. et al. 2010. Origin and Early Development of the Posterior Lateral Line System of Zebrafish. Journal of Neuroscience. 30 (24): 8234–8244.
- Fumey, J. et al. 2018. Evidence for Late Pleistocene Origin of Astyanax mexicanus Cavefish. BMC Evolutionary Biology. 18, article 43.
- For more information on CET, visit ICR.org/cet.
- Borowsky, R. 2018. Cavefishes. Current Biology. 28 (2): R60– R64.
Dr. Boyle is a research scientist at the Institute for Creation Research and earned his Ph.D. in zoology from the University of Hawaii at Manoa.