At a widely publicized news conference in August of 1972, Dr. Jeffrey Bada of Scripps Institute of Oceanography announced the "discovery" of a new dating method based on the rate of racemization of amino acids in fossil material. He was quoted as saying that he had discovered the basis of the method in 1968, and that it was so obvious and simple he was amazed it hadn't been discovered earlier.
As a matter of fact, the basis of this method had been discovered earlier and had been reported in a series of papers published by Hare, Mitterer and Abelson in 1967, 1968, and 1969.1-3 Hare and Mitterer actually reported an estimated age for fossil shell material based on amino acid racemization. 3
Amino acids are the "building blocks," or sub-units, of proteins. About 20 different kinds of amino acids are found in proteins. Each amino acid has two chemical groups, an amino group and a carboxyl group, which can form chemical bonds with other amino acids. The amino group of one amino acid can combine with the carboxyl group of a second amino acid to form a "peptide" bond, and its carboxyl group can combine with the amino group of a third amino acid, and the chain can thus be extended indefinitely. The amino acids combine with each other like the links of a chain to form a long protein chain. Proteins contain from 50 to several hundred amino acids.
All of the amino acids which occur in proteins, except for glycine, which is the simplest amino acid, have at least one asymmetric carbon atom, and can exist as one of two possible stereoisomers. That is, the chemical groups attached to this particular carbon atom are all different and can be arranged in space in two different ways. When there is only a single asymmetric carbon atom, these two different forms are known as optical isomers. Chemically, there is very little difference between them, but biologically, there is as much difference as night and day. The two forms are known as L-amino acids and D-amino acids, the L and D designating the direction in which solutions of these amino acids rotate plane-polarized light. They are mirror-images of each other, and one cannot be superimposed on the other, just as is true of left and right hands.
All amino acids in proteins (except glycine) are L-amino acids. These amino acids spontaneously tend to slowly change to the D-form. The D-form tends to revert to the L-form, and eventually an equilibrium is obtained, as illustrated here for alanine:
Mixture of equal amounts of the L- and D-forms,
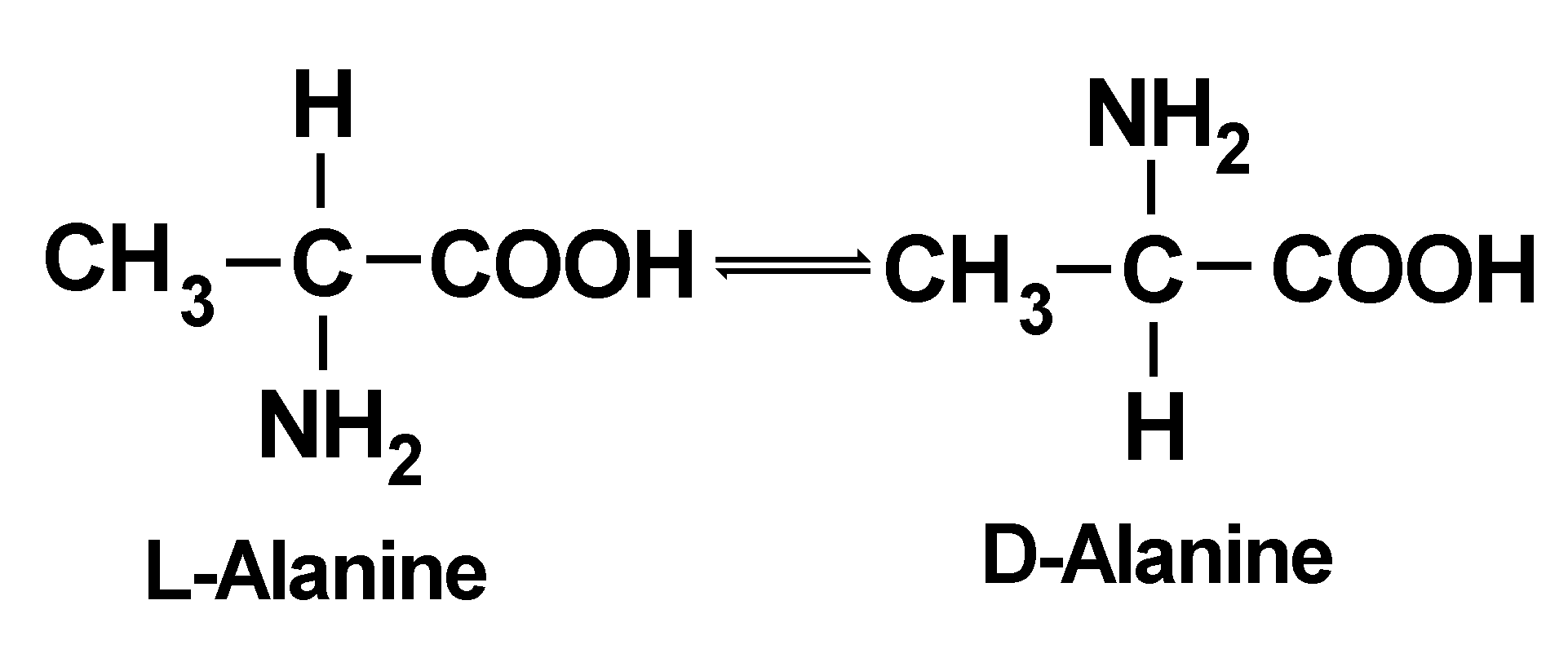 |
The process by which an L-amino acid changes into a mixture of the L- and D-forms (or the D-form changes into a mixture of the L- and D-forms) is called racemization. Racemization is complete when equal amounts of the L- and D-forms are obtained. Complicating things somewhat is the fact that some amino acids have two asymmetric carbon atoms and can exist in four different forms, known as diastereoisomers. Two of these amino acids, isoleucine and threonine, are commonly found in most proteins. L-Isoleucine racemizes (technically in this case, since there are two asymmetric carbon atoms, the correct term is epimerization rather than racemization) almost exclusively to one form, called D-alloisoleucine. Ordinarily it is difficult to separate an L-amino acid from its D-form, but L-isoleucine is easily separated from D-alloisoleucine. The racemization of L-isoleucine to D-alloisoleucine is, therefore, of special interest in the amino acid racemization dating system.
Since the amino acids in proteins of living things are of the L-form, but upon death of the plant or animal spontaneously tend to change to mixtures of the L- and D-forms, the extent of this racemization process could possibly serve as a dating method. Thus, the older a fossil shell or bone, the greater should be the extent of racemization of the amino acids which are contained in the proteins found in the bone or shell.
Hare and Mitterer3 measured the rate of racemization of L-isoleucine to D-alloisoleucine in modern shell fragments heated in water at high temperatures and extrapolated these data to lower temperatures in order to estimate the rate of racemization of L-isoleucine in fossil shells to obtain what they believed to be an approximate age for these fossil shells.
Later, Bada and his co-workers 4,5 reported on their application of the amino acid racemization method for the dating of marine sediments. In other studies, Bada and co-workers have applied this method to the dating of fossil bones, 6-10 and have even applied amino acid racemization rates to the determination of past temperatures by measuring the extent of racemization in several radiocarbon-dated bones.11 Kvenvolden, Peterson and Brown 12 have measured the rates of amino acid racemization in marine sediments. Wehmiller and Hare 13 have also reported on their application of the rate of racemization of amino acids to the dating of marine sediments.
In the study by Bada and Schroeder,5 the rate of racemization in marine sediments under laboratory conditions was determined by heating sediments with sea water in sealed ampoules at various temperatures from 100° C to about 150° C over various lengths of time. The material was then hydrolyzed in 6 molar hydrocholoric acid (the material is hydrolyzed to break up the protein into free amino acids), and the extent of conversion of L-isoleucine to D-alloisoleucine was determined. The rates obtained at these temperatures were extrapolated to 2° C and to 4° C, the present average temperatures where the deep-sea cores containing the sediment samples were obtained.
These data are believed to yield the rates at which L-isoleucine was converted to Dalloisoleucine in the sediment through geological time. The extent of conversion of L-isoleucine to D-alloisoleucine in core sediment samples from various depths was then determined and conclusions based on the above rates were used to estimate the ages of the sediments from various core depths.
The studies carried out with bone were similar.6 Fragments of bone recovered from the Arizona desert and believed to be 2-3 years old were sealed in glass ampoules and heated at various temperatures. The fragments were then hydrolyzed in 6M hydrochloric acid and the extent of racemization of L-isoleucine to D-alloisoleucine was determined. Based on the rates at these elevated temperatures (rates at lower temperatures would be too low to measure), the rates at lower temperatures were estimated. From a combination of conclusions based on these rates, the actual extent of racemization of isoleucine in fossil bones, and the estimated average temperatures at which these fossil bones are believed to have existed, ages were calculated. In other work, the rate of racemization of aspartic acid, instead of isoleucine, was used.8-10
The rate of racemization is highly temperature dependent. The study with bone indicated that an uncertainty of ±2° would yield an age with an error of ±50%. Additional uncertainties are introduced by the possible contamination of the fossil with free amino acids from the environment, and the possibility of racemization during the acid hydrolysis of the protein in the fossil. The former would reduce the apparent age of the fossil by introducing amino acids from recent material which would have undergone little racemization. Racemization which occurs during acid hydrolysis would, of course, increase the apparent age.
Under most circumstances amino acids undergo little racemization during acid hydrolysis, and thus acid hydrolysis is used routinely for the hydrolysis of protein. Under some circumstances, especially effects caused by the nature of the neighboring amino acids, considerable racemization of individual amino acids can occur during acid hydrolysis.14 This necessary step in the preparation of the sample, that is, the hydrolysis of the protein, can itself, therefore, cause the apparent age to be older than the real age.
In amino acid racemization dating methods the above effects, except for the error introduced by uncertainty of temperature, would not ordinarily cause a serious error in the results. There are several factors, however, which the writer believes render amino acid racemization rates useless as a dating method.
Bada and others working in this field have generally assumed that the only two important factors that have influenced the extent of racemization of amino acids in bone, shell, or sediment have been those of time and temperature. It has either been assumed that the nature of the environment has had little influence on the rate of racemization, or that the effect of the environment on the rate has been empirically determined in laboratory experiments.
For example, in the experiments with bone, the rate of racemization was determined in 2-3 year old bone fragments. These data were then applied to fossil bones believed to be several thousand to several million years old. The assumption was, therefore, obviously made that a recent, non-fossilized bone, dried and sealed in a glass ampoule, provides essentially the same environment furnished by a bone undergoing fossilization while standing in soil percolated by groundwater of varying mineral content and of differing pH (the pH is a measure of acidity or alkalinity). This could hardly be the case.
When a bone is deposited in soil, decomposition of the organic material in the bone begins, and the components in the bone undergo a series of chemical reactions with the material contained in the soil. As the organic material decomposes, it is replaced by the minerals contained in the ground water which seeps through the soil. Furthermore, the inorganic material in the bone undergoes change or replacement by minerals contained in the soil. These changes, being a function of the material found in the soil, are irregular, and are governed by the local environment, including mineral content, pH, and temperature. Fossilization, therefore, can occur at greatly differing rates, under circumstances and by processes that vary considerably. The rates of racemization determined by heating dry, fresh bone fragments sealed in glass ampoules could, and most likely would, differ widely from the rates occurring in a bone undergoing fossilization.
Amino acids are especially sensitive to racemization during either the formation of the peptide bond which links the amino acids together, or the breaking of this bond during the hydrolysis of proteins or of peptides (peptides are fragments of proteins of much shorter length than the intact protein). With many years of experience in the synthesis of peptides and in the determination of the structure of proteins, which involves hydrolysis of the protein, the writer can speak from personal experience.
In peptide synthesis, which involves the chemical combination of amino acids in chains of varying length, racemization during synthesis is an ever present concern. Reviews on peptide synthesis always devote special note to this problem.15 Careful choice of reagents, solvents, temperature, and procedure must be made to minimize racemization.
Amino acids, as noted above, are also sensitive to racemization during the breaking of the peptide bond, or hydrolysis. Furthermore, the rate of racemization during hydrolysis is strongly affected by pH. Ordinarily, hydrolysis in strong acid results in little racemization, especially in the absence of impurities. Hydrolysis of a protein in strong alkali, on the other hand, which requires only a fraction of the time required for acid hydrolysis, results in complete racemization of all of the amino acids. Hydrolysis in weak alkali also results in much higher racemization rates compared to hydrolysis at neutral or acid pH. It has been noted that even the rate of conversion of free L-isoleucine to D-alloisoleucine is greatly accelerated in alkaline solution.
It is thus proposed, as has also been suggested by Wehmiller and Hare,13 that most of the racemization that occurs in amino acids of fossil material occurs during the hydrolysis of the protein. It is further suggested that the rate of this hydrolysis, and especially the rate of racemization, is governed mainly by the chemical environment of the fossil material, especially the pH. Temperature could thus play a minor role in determining the extent of racemization. This means that the rate of racemization determined by laboratory experiments under some assumed set of conditions would likely have little or no relevance to the rate of racemization occurring in bone or shell during fossilization.
Local increases in pH, even though temporary, could greatly accelerate the rate of hydrolysis and the rate of racemization, and therefore could result in an apparent age in racemization dating methods vastly older than the real age. Many other chemical effects that occur during fossilization, as yet undetermined, could also have a profound influence on racemization rates. These same general considerations would apply to fossilization that occurs in marine sediments and in other sites.
Bender 16 has recently strongly questioned the reliability of the amino acid racemization dating method. He points out that bones obtained from different levels in the Muleta Cave of Mallorca, when dated by the amino acid racemization method, the radiocarbon method, and by the Thorium-230 method, as reported by Turekian and Bada,7 gave strongly discordant ages. He maintains that amino acid racemization rates are extremely sensitive to the environment. In support, he cited the fact that Kvenvolden and Peterson 17 had found that the extent of amino acid racemization in a supposedly 25,000 year-old bone from a saber-toothed tiger recovered from the LaBrea tar pits hardly exceeded that of modern fresh bone.
Bada,18 in his reply to Bender's criticisms, strongly disagreed that racemization rates in bone are extremely sensitive to the environment. Yet in this same paper, he admits that the results on the material from the tar pits are anomalous, stating (p. 380) that "This type of environment is, however, rare and extreme." These results, nonetheless, conclusively demonstrate that the environment can exert a pronounced influence on amino acid racemization rates. The amino acids in these bones were protected from the environmental influences of soil and groundwater, and consequently suffered practically no racemization. It might be expected, on the other hand, that had these bones been subjected to these environmental factors, the rates of racemization of the amino acids contained in these bones would have far exceeded those obtained in laboratory experiments on bone in the absence of such influences.
There is no doubt that proteins in bone and shell and other fossil material undergo hydrolysis and that the amino acids contained in them suffer racemization with increasing age of fossil material. To use rates of racemization as a dating method, however, the entire history of the fossil material would have to be known, including temperature and the entire diagenetic process, especially the chemical environment that contributed to this process, and most especially the pH. Since all of these factors, most of which accelerate racemization rates, cannot be known, it is suggested that the apparent ages obtained by this method are unreliable and, with few exceptions, are much older than the real ages.
References
1 P. E. Hare and R. M. Mitterer, Nonprotein Amino Acids in Fossil Shells, Yearbook Carnegie Institution of Washington, Vol. 65. p. 362-364 (1967).
2 P. E. Hare and P. H. Abelson, Racemization of Amino Acids in Fossil Shells, ibid., Vol. 66, p. 526-528 (1968).
3 P. E. Hare and R. M. Mitterer, Laboratory Simulation of Amino-Acid Diagenesis in Fossils, ibid., Vol. 67, p. 205-208 (1969).
4 J. L. Bada, B. P. Luyendyk, and J. B. Maynard, Marine Sediments: Dating by the Racemization of Amino Acids, Science, Vol. 170, p. 730-732 (1970).
5 J. L. Bada and R. A. Schroeder, Racemization of Isoleucine in Calcareous Marine Sediments: Kinetics and Mechanism, Earth and Planetary Science Letters, Vol. 15, p. 1-11 (1972).
6 J. L. Bada, The Dating of Fossil Bones Using the Racemization of Isoleucine, ibid., Vol. 15, p. 223-231 (1972).
7 K. K. Turekian and J. L. Bada, The Dating of Fossil Bones, in Calibration of Hominoid Evolution, ed. by W. W. Bishop and J. A. Miller. The Wenner-Gren Foundation for Anthropological Research, New York, 1972, p. 171-185.
8 J. L. Bada and R. Protsch, Racemization Reaction of Aspartic Acid and Its Use in Dating Fossil Bones, Proceedings National Academy of Science, Vol. 70, p. 1331-1334 (1973).
9 J. L. Bada, K. A. Kvenvolden, and E. Peterson, Racemization of Amino Acids in Bones, Nature, Vol. 245, p. 308-310 (1973).
10 J. L. Bada, R. A. Schroeder, and G. F. Carter, New Evidence for the Antiquity of Man in North America Deduced from Aspartic Acid Racemization, Science, Vol. 184, p. 791-793 (1974).
11 R. A. Schroeder and J. L. Bada, Glacial-Postglacial Temperature Difference Deduced from Aspartic Acid Racemization in Fossil Bones, ibid., Vol. 182, p. 479-482 (1973).
12 K. A. Kvenvolden, E. Peterson, and F. S. Brown, Racemization of Amino Acids in Sediments from Saanich Inlet, British Columbia, ibid., Vol. 169. p. 1079 (1970).
13 J. Wehmiller and P. E. Hare, Racemization of Amino Acids in Marine Sediments, ibid., Vol. 173. p. 907-917 (1971).
14 J. M. Manning and S. Moore, Determination of D- and L- Amino Acids by Ion Exchange Chromatography as L-D and L-L Dipeptides, Journal of Biological Chemistry, Vol. 243, p. 5591-5597 (1968).
15 See for example Duane T. Gish, Peptide Synthesis, in Protein Sequence Determination, ed. S. B. Needleman, Springer-Verlag, New York, 1970; Peptides, ed. L. Zervas, Pergamon Press, New York, 1966, p. 121-155; E. Schroder and K. Lubke, The Peptides, Vol. I, Methods of Peptide Synthesis, Academic Press, New York. 1965, p. 323-325; M. Bodanszky and M. A. Ondetti, Peptide Synthesis, Interscience Publishers, New York, 1966, p. 18-19.
16 M. L. Bender, Reliability of Amino Acid Racemization Dating and Paleotemperature Analysis on Bones, Nature, Vol. 252, p. 378-379 (1974).
17 K. A. Kvenvolden and E. Peterson, Amino Acids in Late Pleistocene Bone from Rancho LaBrea California, Geological Society of America Abstracts with Programs, 1973, p. 704 (see also Ref. 9).
18 J. L. Bada, Dr. Bada Replies, Nature, Vol. 252, p. 379-381 (1974).
* Dr. Gish is Vice President of ICR.






