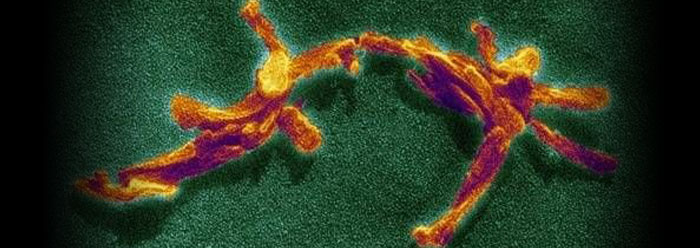
Medical students learn classic pathology cases to help them identify diseases and their causes. One such case involved human cannibals who ended up with tremors, seizures, balance disorders, and hallucinations after eating nervous tissue. The fatal diseases, Kuru disease and Creutzfeld-Jakob disease (CJD), which are akin to mad cow disease, are caused by mysterious transmissible proteins known as prions. These proteins can be found in neurons. When a prion becomes pathologically misshapen, an infectious change reaction occurs where prions in the host become shaped in the same way as the infecting prion. Buildups of damaging amyloid and tiny holes in the brain at the site of disease follow. Similar illnesses in animals like chronic wasting disease in deer and mad cow disease in cattle are also spread by prions.
Prions pass traits by their shape, not DNA
In spite of their gloomy notoriety, prions are fascinating to study. Prions reveal another way information is transmitted between individuals and even across generations. They pass heritable traits from cell to cell by their self-patterning structure—they literally folded in specific ways—instead of using DNA.
Researchers at Stanford University Medical School investigated proteins in yeast cells capable of passing on traits for at least 100 subsequent generations.1 The goal was to study the range and duration of the inheritance of traits produced by proteins coded for by this yeast's 5300 genes. For 48 hours the research team induced each gene to overexpress from 10 to 100 times more protein product.
Their research yielded at least four new discoveries. First, the transient overexpression experiment found 46 proteins with prion-like behavior. Inheritance patterns matched those of typical prions since all daughter yeast cells inherit the entire prion repertoire from a parent cell, and the prion-induced traits become dominant.
Second, when the team analyzed the prions' shapes, only a few of them resembled usual prions that fold in a way that formed long fibrils packed tightly together. Most of the newly discovered prions lacked that trait. Instead, an "intrinsically disordered" domain enabled them to fold in a wide variety of ways. Yet one common feature was that all prions shared an attraction to DNA molecules.
Third, the ability of most of these proteins to assume different shapes enabled the yeast to have novel traits which, remarkably, continued to be heritable for many generations even after their expression returned to normal ranges. Finally, the traits modified the capability of the yeast in certain conditions such as enhancing resistance to anti-fungal drugs or growth in higher temperature ranges.
Evolutionists claim prions enable survival of the fittest
Stanford University's press release detailed publication of this research. It identified the scientists, disclosed significant funding, and claimed that the "work is an example of Stanford Medicine's focus on precision health, the goal of which is to anticipate and prevent disease in the healthy and precisely diagnose and treat disease in the ill."2 However, no significance of the work's benefit to public health was ever identified, but its perceived importance to evolutionary theory was. It quoted one author's take on the implications of the research,
"In evolution there's a paradox," said Daniel Jarosz, Ph.D., assistant professor of chemical and systems biology and of developmental biology, who is lead author of the study. "We know that there are an extraordinary number of mechanisms that exist to protect the integrity of the genetic code and to assure that it's faithfully passed on to future generations. But we also know that evolutionary success requires adaptability. How can you reconcile that need with the fact that the raw material for that innovation is really limited?"
The research paper published in Cell provides the answer. In this case, adaptability does not depend on "limited" genetic mutations since, "our data thus establish a new and common type of protein-based molecular memory through which intrinsically disordered proteins can drive the emergence of new traits and adaptive opportunities."1
The close similarity between these new prion-like proteins and human proteins with prion-like characteristics (and their underlying genetics), which both enable the physical flexibility to fulfill a variety of roles in a cell, was not missed. They interpret these prion-induced traits that enhance performance in certain conditions as a Darwinian survival advantage. Even though yeast and human evolution is claimed to be separated by vast eons, the paper asserts that "these domains have been widely conserved across evolution, and several human homologs had the capacity to fuel protein-based inheritance."1 The writer for PhysOrg says that actually they "were conserved over hundreds of millions of years" supposing that, according to coauthor Chakrabortee, "This conservation over millennia could be because these proteins are vastly beneficial in nature."3
A design-based approach to frame the findings
One key question is: How do yeast produce an apparently suitably targeted trait after being exposed to a stressful condition—not using iterative rounds of trial and error? Neither the research paper nor the press release have many details that address this question. The press release touches on the topic by noting that after yeast cells become stressed by sunlight "they begin producing large quantities of proteins called molecular chaperones—or proteins that help other proteins fold. These chaperones wrap around prions and fold them into a shape that kicks off a chain reaction of sorts. Other prions of its kind follow suit, using the original as a template."2
Sunlight itself does not form chaperone proteins. What is the link between sunlight exposure and yeast making chaperones? Also, the prion shape is linked to a new or altered trait enabling the yeast to self-adjust specifically to more sunlight. The press release states, "Chaperones wrap around prions and fold them into a shape." The report is somewhat misleading since the glib statement that chaperones fold prions into "a shape" is not just any shape, but a specific shape. Knowing the underlying causes for these types of specification is fundamental to understanding function. What determined the selection of a specific shape? How is the information for the logic controlling the internal selection event and the prion shape accessed and applied?
What if a purposely designed unified system controls the detection of sunlight, the direction of prion folding, and the expression of specific traits? It is likely that the yeast is equipped with a sensor specific for light, and a mechanism happens to relay that data internally where if-then logic (i.e., if sunlight-then make chaperones) to produce chaperones. These chaperones fold prions into specific shapes that kick off downstream events leading to specific traits. None of this happens via Darwinian mechanisms since information is passed on by shape and not genetics, and given the lack of trial-and-error iterations. Therefore, intelligent design is a more logical explanation for all of this data—it's a better fit.
It is also reasonable to infer that common design explains why the mechanism is the same in diverse organisms. Evolutionary invocations of "conservation" are not observations demonstrated to flow from objectively-discernable causes. Conservation is actually a declaration which presupposes evolution—none of the researchers actually observed conservation happening. Conservation is based on mental pictures of diverse organisms evolving in diverse environments that somehow serendipitously preserve some DNA unchanged, while the other DNA mutates all around it.
Regardless of interpretation, protein-based inheritance via intrinsically disordered proteins does lead to new adaptive traits "that in yeast, and possibly other organisms, including humans" is "more widespread than previously believed...."4 The study rightly concludes that "Prions are a paradigm-shifting mechanism of inheritance in which phenotypes are encoded by self-templating protein conformations rather than nucleic acids."1
Intentional design is a far better explanation for this mechanism being the same in diverse organisms than trial-and-error evolution.
References
- Chakrabortee, S., et al. 2016. Intrinsically Disordered Proteins Drive Emergence and Inheritance of Biological Traits. Cell. 167 (2): 369–381.
- Spector, R. Prions can pass on beneficial traits, study finds. Stanford Medicine News Center. Posted on med.stanford.edu October 3, 2016, accessed October 18, 2016.
- Revising the meaning of 'prion.' Phys.org News. Posted on phys.org October 4, 2016, accessed October 18, 2016.
- Prions can pass on beneficial traits. ScienceDaily. Posted on sciencedaily.com October 3, 2016, accessed October 18, 2016.
Image credit: Copyright © Eye of Science/Science Photo Library. Adapted for use in accordance with federal copyright (fair use doctrine) law. Usage by ICR does not imply endorsement of copyright holder.
* Dr. Guliuzza is ICR's National Representative.
Article posted on November 3, 2016.