If you have stopped receiving emails from us, please check your SPAM folder.
Also, to help stop emails from getting mistagged as spam in the future,
be sure to add 'webmaster@my.icr.org' to your contact list!
Click here for more tips!
Also, to help stop emails from getting mistagged as spam in the future,
be sure to add 'webmaster@my.icr.org' to your contact list!
Click here for more tips!

Moroccan Dinosaurs in Marine Rocks, Too
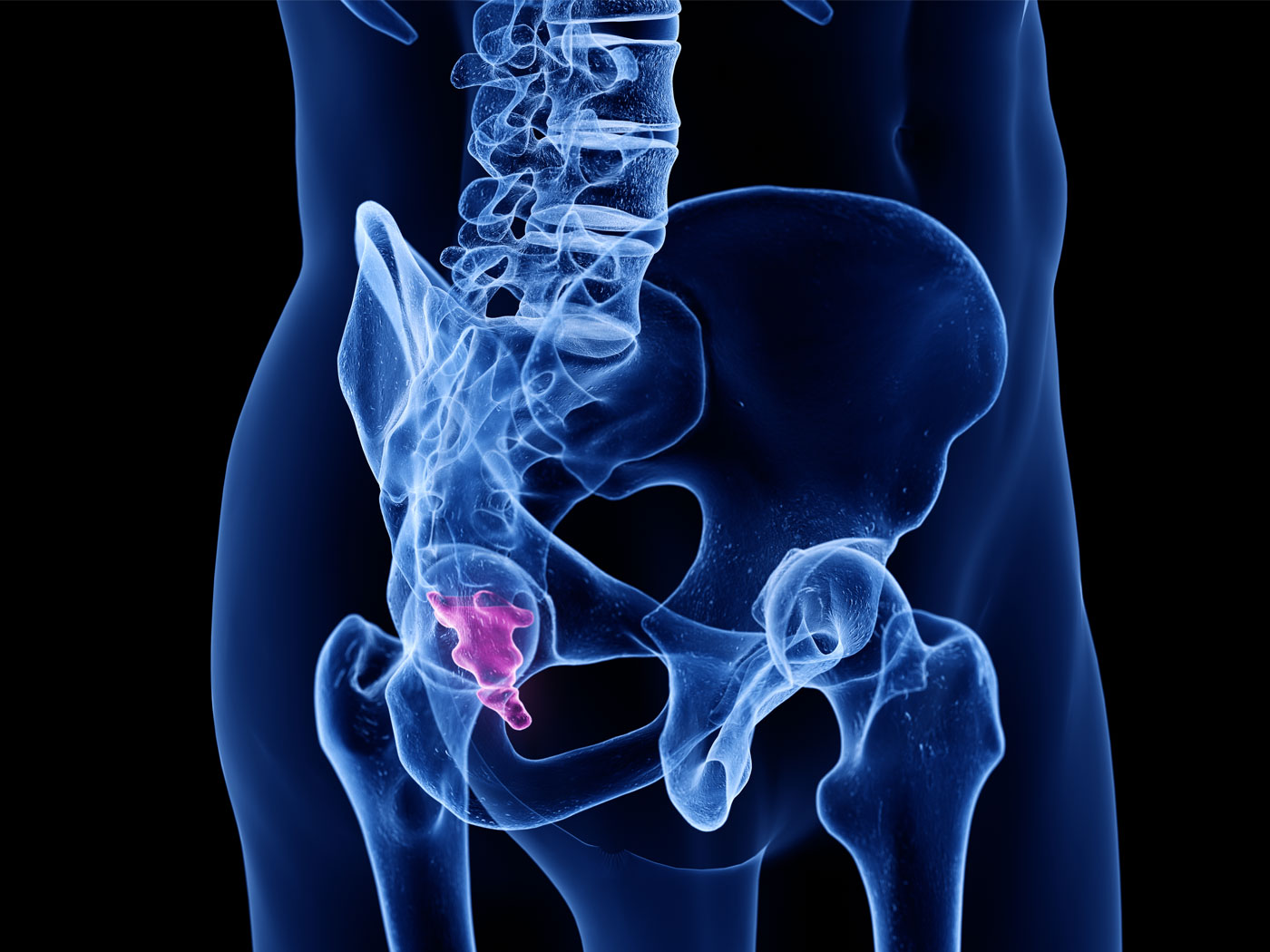
Two recent papers by paleontologist Nicholas Longrich and his colleagues describe some unexpected findings in phosphate mines of northern Morocco.1,2 Most surprisingly, they found a pair of short-armed carnivorous dinosaurs mixed in with fish, sharks, and marine turtles in the same layer.1 Their second discovery was a 26-foot-long, previously unknown mosasaur with “bladelike” teeth that was also found in the same formation as the dinosaurs and marine creatures.2 Why are dinosaurs and mosasaurs in the same marine rocks?
Located in the Khouribga...
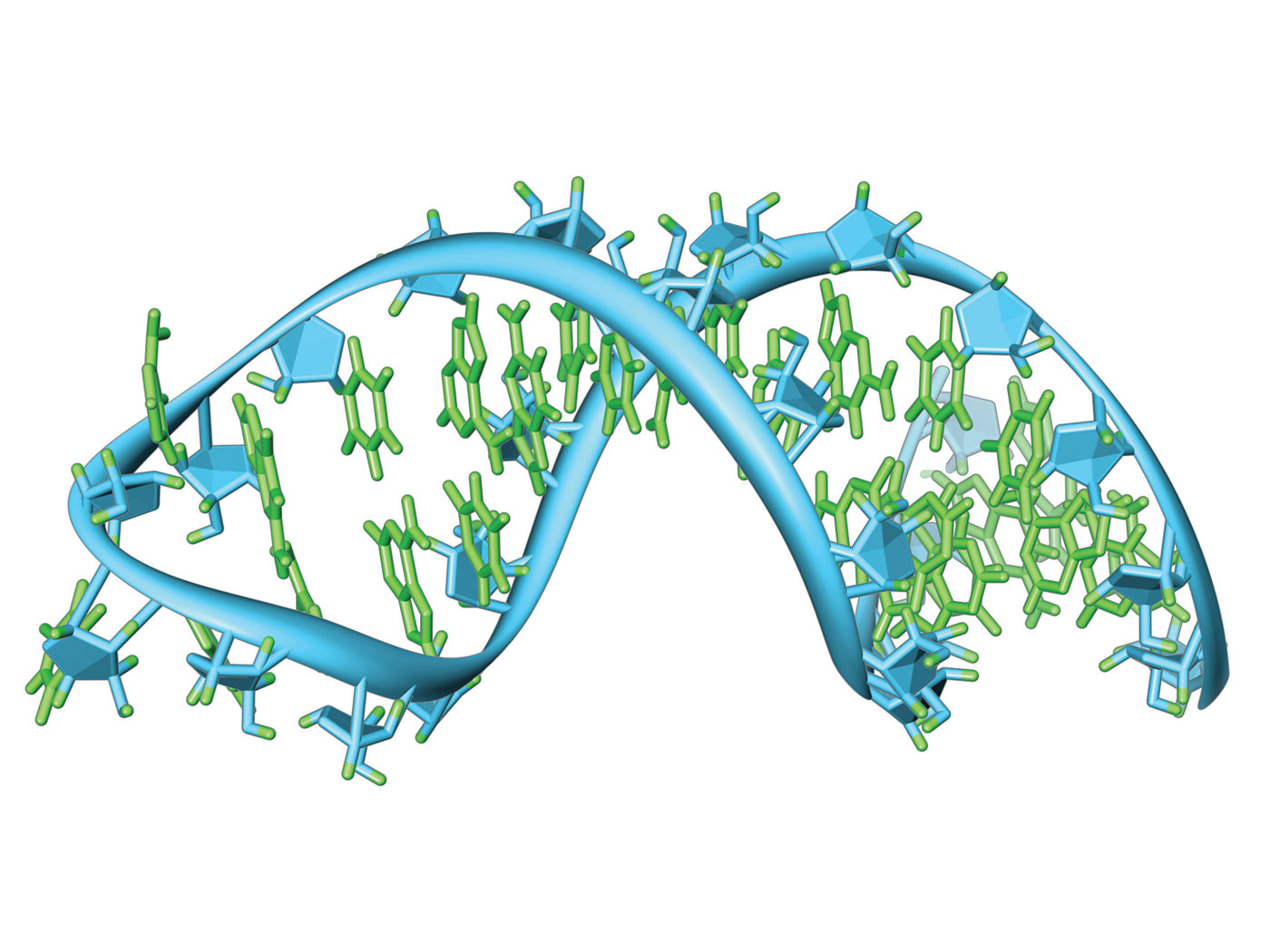
RNA Editing: Adaptive Genome Modification on the Fly
When the workings of the genome were first being discovered, the central evolutionary dogma of molecular biology claimed that genetic information passes consistently from DNA to RNA to proteins. Today we know that RNA...
The Latest
Moroccan Dinosaurs in Marine Rocks, Too
Two recent papers by paleontologist Nicholas Longrich and his colleagues describe some unexpected findings in phosphate mines of northern Morocco.1,2...
CREATION PODCAST
Ernst Haeckel: Evolutionary Huckster | The Creation Podcast:...
Ernst Haeckel, a German Zoologist, is famous for developing a series of images of embryos in development called Anthropogenie. These images,...
Bees Master Complex Tasks Through Social Interaction
Bees are simply incredible.1,2 These little furry fliers challenge the very foundation of Darwinism in many diverse ways.
Bees have been...
The Tail of Man’s Supposed Ancestors
Although it has been known for decades and despite insistence to the contrary from the evolutionary community, man—Homo sapiens—has never...
When Day Meets Night—A Total Success!
The skies cleared above North Texas on Monday, April 8, for a spectacular view of the 2024 Great American Solar Eclipse. Hundreds of guests joined...
The Sun and Moon—Designed for Eclipses
Before discovering thousands of planets in other solar systems, scientists tended to assume that other solar systems would be very similar to our own....
Let ICR Help You Prepare for the Great American Solar Eclipse!
On Monday, April 8th, the moon will move directly between the earth and the sun, resulting in a total solar eclipse visible in northern Mexico, much...
Total Eclipse on April 8th
“You alone are the LORD; You have made heaven, the heaven of heavens, with all their host, the earth and everything on it, the seas and all that...
CREATION PODCAST
Dismantling Evolution One Gear At A Time! | The Creation Podcast:...
The human body is a marvel of complexity and the more we learn about it, the more miraculous our existence becomes! Can evolution explain the...
April 2024 ICR Wallpaper
"He appointed the moon for seasons; The sun knows its going down." (Psalm 104:19 NKJV)
ICR April 2024 wallpaper is now available...
Featured Event
Kids on Mission
Video
We Are More Than Animals! #creationism #podcast
Ernst Haeckel: Evolutionary Huckster
Penguin Party: Surviving Extreme Cold
An Imaginary Theory #podcast #creationism
More in Video ⊳